Cation exchange chromatography Elution buffer: 0.05 M Tris-HCl ( pH... | Download Scientific Diagram
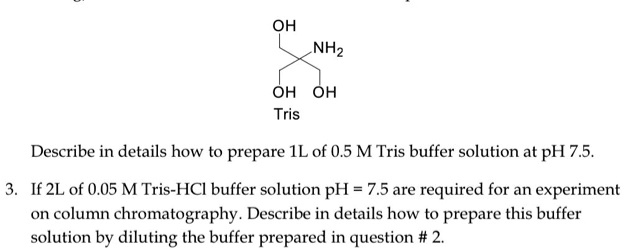
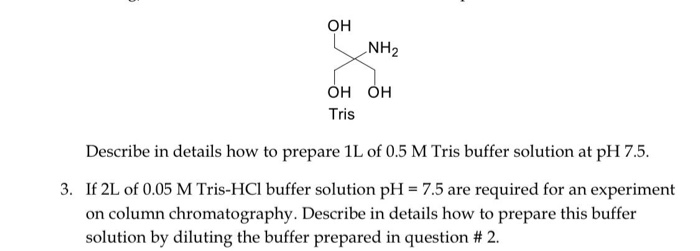
SOLVED: OH NH3 OH OH Tris Describe in detail how to prepare an IL of 0.5 M Tris buffer solution at pH 7.5. If 2L of 0.05 M Tris-HCl buffer solution with

TNT Buffer (0.1M Tris.HCl pH 7.5, 0.15M NaCl, 0.05% Tween®-20) - Cepham Life Sciences Research Products

SOLVED: How would you prepare 100 mL of a 50 mM Tris-HCl buffer at pH 7.4, using the 0.5 M Tris base stock solution and any other necessary materials?